ORP分析仪-ORPSense

ORP传感器
Pi公司的ORPSense系列ORP分析仪是当今世界上最先进和最好的ORP传感器,可用来在线测量任何溶液的氧化还原电位。它们是基于铂电极的传感器,内置参比电极,不使用任何试剂,非常稳定,并且大大减少了维护量,降低了整个使用周期的成本。
在线ORP仪和ORP控制器是目前在水处理中最常用的分析仪器之一,但在线ORP仪表经常给操作人员带来很多问题。我们需要的是一个稳定、可靠、成本合理的在线ORP仪表。这正是ORPSense和ORPSense自动校准器的功能。
ORPSense传感器和流通池可与不同的控制器一起使用,通过不同的通信、显示和控制选项,为您带来出色的性能
Pi公司的pH/ORP计的核心部件是一种具有专利的聚合物填充玻璃电极。这种ORP电极比市场上许多的ORP电极都要好得多,寿命长,漂移小,可靠性高。与许多市面上的ORP分析仪不同的是,你不需要搜索数以百计的ORP电极,以找到一个适用你的应用的电极。ORPSense系列传感器的电极适用于pH值2到12、导电率为0µs以上的所以应用。虽然它们的价格较高,但这些传感器的使用寿命更长,维护要求也低得多,成本效益更高,通常只需每两三个月校准一次。
尽管包含了所有的附加功能,购买成本却低于其竞争对手! ORPSense系列ORP分析仪使用非常稳定的ORP电极,因此无需对ORP分析仪进行持续和重复地校准。ORPSense会计算出“置信度”,并让你知道它的置信度是否很低。如果ORP计检测到ORP传感器出现故障,它就会在显示器上给出指示,说明可能出现的故障,并进行全面的故障诊断。
在线ORP分析仪能够进行全PID控制,并配备了过程控制选项、数据记录、继电器输出、串行通信(TCP、以太网、Modbus、Profibus)。ORP分析仪可通过internet和LAN进行远程访问(包括对所有控制选项的远程访问)。事实上,ORPSense拥有你想要的所有ORP仪表选项,同时保持低成本和高性价比。
- 稳定可靠的ORP仪表
- 在线诊断
- 采购成本低
- 长寿命
- 远程访问和短信报警
- PID和其他过程控制
- 偏远用户

ORP 传感器
- 冷却塔
- 食物生产
- 造纸
任何需要测量ORP的地方都适合ORPSense。ORPSense 系列的ORP计特别适合在可靠性和易用性要求高的场合工作。
ORPSense系列在线ORP监测仪可以配备自动清洗装置。用户可定义冲洗的时间间隔。自动清洗装置在食品、纸浆和造纸中特别有用,在许多可能会在样品中沉积固体的应用中也很有用。
ORPSense系列在线ORP监测仪可与其他传感器配套组成多参数系统。详情请联系您当地的经销商。
文件名 | 文件类型 | 大小 |
|---|---|---|
手册 | 982kB | |
手册 | 678kB | |
技术说明 | 553kB | |
手册 | 935kB | |
手册 | 939kB | |
手册 | 975kB | |
技术说明 | 649kB | |
技术说明 | 593kB | |
手册 | 656kB | |
技术说明 | 316kB |
聚焦专题(Focus On)是通过电子邮件分发的一系列短文,提供有关仪器仪表和饮用水、废水、工艺用水、泳池水的过程测量的技术信息。如果您想加入收件人列表,请与我们联系。
余氯通常需要被从工艺中去除,原因有很多。氯可能会损坏反渗透膜,或使食物变成难看的棕色。除氯对仪表制造商和工程师们提出了一些挑战,但是…
…你知道ORP可以有效控制亚硫酸氢钠的投加量吗?
…你知道ORP可以限制亚硫酸氢钠的过量投加(这会导致以后的工艺中出现问题)吗?
…你知道ORP对氯含量非常低时的变化非常敏感吗?
如何有效地控制余氯的去除?
从逻辑上讲,使用一个标准的余氯探头并投加还原剂(如亚硫酸氢钠),直到读数归零,是完全合理的。如果读数从零开始增加,则应使用更多的还原剂。
不幸的是,由于余氯传感器固有的电化学特性,这种完全合乎逻辑的解决方案将不起作用。这是因为安培法余氯探头需要一些氯才能正常工作。这通常被称为极化,探头在没有氯的水中停留的时间越长,在重新加氯时,检测到氯所需的时间就越长。这不是由于制造质量或设计缺陷。这是一个基本的电化学性质,很难突破。

余氯传感器头部
一些安培法探头比其他探头更能抵抗这种影响。它们适用于余氯可能间歇性达到零的应用,或者可以在消毒后的水中(通常是饮用水)定期复位的应用。Pi的HaloSense Zero传感器就是在考虑到这一点的情况下制造的。这种安培零余氯传感器已成功地应用于反渗透膜和许多其他低氯工艺中。
然而,在使用还原剂去除氯的应用中,一种更优雅的解决方案是使用ORP。ORP传感器测量氧化还原电位。

ORP传感器
氯与还原剂之间的反应是还原氧化反应。这意味着ORP可用于控制这一过程,并可确保去除氯,同时防止还原剂过量。
下面是HOCl和NaHSO3之间反应的简化方程式。
2NAHSO3 + 2HOCl → H2SO4 + 2HCl + Na2SO4
当游离氯过量时,ORP升高,当亚硫酸氢钠过量时,ORP降低。
在低浓度下,氯浓度的微小变化将导致ORP的大变化。这是因为ORP与氧化剂和还原剂的浓度之间的关系是对数的。还原剂(如亚硫酸氢盐)的浓度也是如此。
Pi的ORPSense可以用来控制氯的去除。ORP滴定曲线是选择ORP设定值所必需的。该图显示了一个典型的ORP滴定曲线,尽管每个工艺的准确值不同。重要的是要测试每一个新安装的传感器,并相应地调整ORP设定值。一旦确定了滴定曲线,ORP传感器就不需要进行频繁的校准。
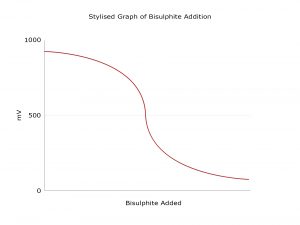
值得一提的是,ORP受工艺水中许多其他变量的影响,如电导率、pH值或温度,并且这种控制方法并不适用于所有工艺。结合Pi的CRONOS®或CRIUS®控制器,Pi的ORPSense可以与其他传感器一起安装,如零余氯传感器、pH或电导率传感器,帮助您更完整地控制您的工艺流程。
如果你需要控制一个用化学物质、碳过滤器甚至紫外线去除氯的工艺,Pi可以通过提供世界级的仪表和专业知识来帮助你。
注意,这个Focus On话题讲述的是关于氯的去除,然而,以上的许多原理可以推及其他氧化剂,如二氧化氯。
你可能知道,一些人使用ORP来控制氯的投加,而另一些人使用ppm余氯传感器,但是…
你知道ORP超过3ppm是测不准的吗?
你知道美国的游泳池使用ORP,欧洲的游泳池使用ppm余氯传感器吗?
你知道城镇的水的ORP会有很大的变化吗?
在美国,几乎所有的游泳池和spa都使用ORP传感器来控制氯的投加,而在英国和西欧,大多数ORP系统已经被测量水中游离氯浓度的系统所取代。Pi提供使用其中一种或两种技术的系统。
ORP
氧化还原电位(ORP或氧化还原)传感器,测量水从水中的任何东西获得或失去电子的倾向。ORP的读数越高,水氧化(从水中的有机物或其他物质中获得电子)的趋势越强,从而杀死或摧毁它们。
为什么在美国这么多的游泳池使用ORP?

游泳池
当氯被加入池中时,它会形成OCl-和HOCl。消毒主要由HOCl完成,ORP对HOCl在水中的浓度做出响应,这使得它可以很好地衡量水中氯杀死细菌的趋势。尽管如此,ORP是HOCl的一个次要度量,并且受到许多其他因素的影响。下面将讨论其中一些因素。ORP的主要优势是:采购成本低,无须校准,很少或无须维护。
ORP传感器有什么问题?

ORP传感器
不幸的是,ORP传感器测量的是趋势而不是能力,即ORP测量的是水杀死细菌的可能性或能力,而不是水能杀死多少细菌,这是一个微妙但非常重要的区别。一个高ORP的水样可能很快就能杀死少量的细菌,但却不能杀死未来的污染。更重要的是,虽然氯对ORP的影响非常强烈,但它并不是唯一的变量。水的pH值直接影响ORP,同时也影响两种主要消毒剂OCl-/HOCl的浓度比。较低的pH值(较高的酸度)会导致HOCl相对浓度的增加,从而导致ORP的增加。
ORP最大的问题可能是没有氯的水的ORP读数会因水的来源不同而不同。这意味着,两个地区的ORP浓度同样为750mV,而余氯浓度却可能不同。此外,ORP对HOCl的响应不是线性的,将余氯增加到3ppm以上对ORP读数的影响很小,在3ppm以上的控制变得非常困难。这些问题通常会导致过量的加氯。这一点在美国的余氯含量经常超过2 ppm的泳池中可以很明显地看出来,而欧洲泳池的余氯含量通常在0.8-1.5 ppm之间(世界卫生组织建议的残留量为1 ppm)。
ppm余氯
这些传感器使用电化学法直接测量游离氯浓度。它们往往比ORP传感器稍贵一些,但更具有可重复性和准确性,因此往往具有更好的控制能力(因此降低了化学成本)。它们是针对游离氯(消毒剂)的,可以很容易地使用DPD测试来校准游离氯的浓度。虽然ppm余氯传感器的价格较高,但总体的成本往往较低,因为ORP传感器通常每年更换一次,而ppm传感器可以使用10年或更长时间。
ppm氯传感器的问题
一个ppm传感器测量的是水杀死有机物的能力,唯一的问题是它不能测量细菌被杀死的速度,一个很大程度上取决于pH值的变量。ppm传感器有两种不同的类型。一种传感器只测量HOCl,和ORP传感器有非常相似的问题。另一种传感器,在pH低于8.0时,同时测量HOCl和OCl-。Pi只建议使用不受pH值影响的传感器。这就导致了对pH值和游离氯的更严格的控制,这意味着可以更严格地控制和减少余氯,从而降低成本,并获得更愉快的沐浴体验。
结论
| 优点 | 缺点 |
|---|---|
ORP传感器简单(无须校准) | ORP传感器不测量消毒能力 |
ppm传感器直接测定游离氯 | ppm传感器需要校准 |
在整个水处理工业中,由于积垢,许多应用很难保证仪表每天都正常工作。但是你知道吗…
…针对大多数类型的传感器,Pi公司都配备了自清洗和自冲洗系统?
…这些污垢清洗系统可以延长传感器的寿命,并大大减少维护量?
…Pi的自清洗/冲洗系统设计合理、简单且无故障?
问题是什么呢?
传感器积垢

无论被监测的是何种工艺,在样液中经常有一些东西能够污染传感器,从而导致错误的读数。解决这一问题的办法是清洁传感器,但每种仪器的检查和清洁程序应如何设定?检查和清洁过于频繁,既费时又费钱。不够频繁,仪器会给出错误的结果,并可能过早地达到寿命。
有什么解决方案?
Pi公司的Autoclean 系统和 Autoflush系统。
Pi公司的自动清洗/自动冲洗系统简单、可靠且易于维护,可替代机械清洗方法,机械清洗方法会导致堵塞和破损。通过定期向传感器/探头喷出干净的水或空气,传感器在很长一段时间内保持清洁且无污垢。传感器清洗周期可通过Pi的控制器设定,用户可选择冲洗的时间和频率,无论应用程序有多脏,探头都能保持清洁。传感器本体或清洁附件中没有活动部件,除了一个简单的阀门,没有任何东西需要更换或检查。
Pi的自动清洗和自动冲洗系统可以使传感器无故障、无污染地工作数周,甚至数月。
针对不同的应用的不同的解决方案
Autoclean自清洗系统

这个选项可以添加到我们的pH,ORP,浊度,悬浮固体和溶解氧(DO)传感器上。包括一个端盖,干净的水(或空气)流过传感器表面,以清除任何污垢。清洁由一个阀门控制。
Autoverify自检系统
如果使用空气来清洗溶解氧传感器,系统还可以自动验证传感器是否仍然正确响应。避免了经常取出传感器检查的麻烦。
Autoflush自冲洗系统

对于诸如余氯、臭氧和二氧化氯等需要安装在流通池中的传感器,Autoflush自动冲洗系统可通过内置的阀控制样液和并清洁水的进出和流动。用户可以设置冲洗间隔和持续时间,以确保传感器没有污垢。对于特别脏或顽固的污染物,可用温水作为冲洗水来帮助清洁。
有了上述选项,无论测量的应用或参数是什么,Pi公司都能够提供这样一个监测系统:不仅精确和稳定,而且还将保持无污染,节省操作人员的时间和金钱。
“来自Pi及其合作伙伴的支持是一流的。他们不仅要确保他们的设备完美,而且要确保整个过程也很好。五星!”
Anthony Glitto
Equip Solutions-美国伊利诺斯州
“英国Pi公司总有一个高水平的客户服务。我们与Pi的所有互动都超出了我们的预期。和你们一起工作总是一件很愉快的事。”
Rudi Tuffek
Allpronix -南非
“从ORP控制到安培法余氯传感器控制,无疑已经极大地改善了池水水质!”
Chris Tedeschi
Chris Tedeschi
“为客户服务不仅仅是解决问题或解决投诉,Pi非常胜任这些。他们通过技术和快速的努力提供良好的体验。”
Clovis Tuchapski
Buckman -拉丁美洲
“Pi的产品物有所值,代表了最好的市政饮用水分析仪。”
John Clark
Chemtrac –美国亚特兰大
“在过去的几年里,我们从Chemtrac购买了余氯和浊度分析仪,通过常规校准,探针准确测量了余氯和浊度,没有任何问题。我们对这个产品非常满意,并强烈推荐他们。”
Daniel "Buck" Owen
Ocoee公用事业区
“我们ECM ECO监控部门只能向所有其他潜在客户推荐Pi的产品和服务。 他们拥有非常复杂的各行业水质监测产品组合,态度友好,交货时间非常快,并及时响应我们所有的需求和问询。 我们的客户特别感谢颗粒计数器,它们帮助识别饮用水处理问题。流动电流监测器是优化昂贵化学品的一个很好的工具。”
Branislav
ECM ECO Monitoring-斯洛伐克











